Key-Words- Fungal keratitis, Cornea, Vegetative trauma, Contact lens, Diabetes mellitus, Corticosteroid
INTRODUCTION- Eye is one of the important organs of sensory per-ception. Normally it is kept free from microbial in-fections by the natural protective mechanisms. Breach of these protective barriers can predispose to ocular diseases leading to significant ocular morbidity. Keratitis as such is an in-flammation of the cornea, due to infectious, physical or chemical causes, which can result in vision loss. Contact lens use is the main predisposing factor for infectious keratitis [1]. Eye is also vulnerable to infections with fungal members due to trauma and excessive steroid use with or without antibiotic. There has been increasing trends of infectious corneal blindness owing to higher incidences of direct ocular trauma added with personal and social factors like increased alcohol consumption, higher age, low socioeconomic status and lack of health education.
[2-3] Fungal keratitis is one of the major causes of the ulcerative and sight threatening infection of the cornea but its incidence is usually underestimated in the community.
Fungi are ubiquitous eukaryotic microorganisms. Fungi that infect the cornea are broadly classified as yeast or moulds. Yeasts are unicellular fungi characterised by an oval or round structure, the blastoconidium. Moulds are organisms with filamentous structure called hyphae and a tangled mass of hyphae which constitutes the mycelium. Filamentous fungi may be classified as septate or non-septate. Fungi reproduce sexually by the formation of spores and asexually by forming conidia or sporangiospores. The fungi responsible for keratitis are cultured in asexual phase most of the times. Fungi are not capable of infecting the normal intact cornea and subsequent injury or defect is needed for fungal inoculation into healthy epithelium. As such they are only capable of causing keratitis once gain access to the cornea through defect. Aspergillus and Fusarium are among the common causes of fungal keratitis worldwide. Aspergillus generally having the worst prognosis, but it does show a good response to antifungal drugs. [4]
Diagnosis of fungal keratitis possesses a clinical chal-lenge and requires a very high degree of clinical suspicion. The traditional direct microscopic examination of cornea is still an easy and handy clinical tool for diagnosis. [5] Fungal keratitis can be managed with antifungal drugs without steroids. The predominant antifungal implicated in management of fungal keratitis includes natamycin and voriconazole [6]. Collagen cross linking with antifungal agentsÆ introduction in stroma of cornea has a promising outcome in fungal keratitis [7]. Therapeutic corneal transplant is an effective alternative for refractory fungal keratitis despite complications associated with it, like recurrence of infection and graft failure [8]. There is also convincing evidence of hydrogel based contact lenses and drug delivery system to decrease the contact lens induced fungal keratitis [9].
Fungal keratitis can lead to complications like hypo-pyon formation and even sight threatening endophthalmitis, if untreated. Fungal keratitis can also lead to local complication like fungal scleritis [10]. It is a significant cause of mono-ocular blindness in some countries [11]. The study regarding the true prevalence and incidences of fungal Keratitis with its related morbidity is lacking. The ob-jectives of this study were to determine its incidence and associated etiological risk factors in this region.
MATERIALS AND METHODS- A prospective case study of patients presenting with clinically suspected keratitis was conducted at Al-Rehma Hospital, Sirte, Libya between January 2008 and November 2010. All the patients presenting with clinical features of keratitis during the specified time period were included in the present study.
Case definition: Keratitis was defined as infiltration of the corneal epithelium with or without stromal infiltration, suppuration or hypopyon. Ulcers with typical features of viral infection, Mooren's ulcers, interstitial keratitis, sterile neurotrophic ulcers, and other ulcers associated with autoimmune conditions were ex-cluded. A standardised form was filled out for each patient to document detailed history including so-ciodemographic data, the mode of onset, the traumatising agent, duration of symptoms, previous and current ocular or systemic disease and ophthalmic or systemic medication.
Techniques: Each patient was examined on slit lamp by the ophthalmologist. The defect size in epithelium was measured by staining with 2% fluorescein. Measurement was carried out by slit lamp microscopy using different slits and results interpreted in millimetres. This was followed by measuring the extent of stromal infiltration and drawing the sketch in reference to standard frontal and cross sectional dia-grams. The presence or absence of associated ocular conditions such as lid abnormalities, Bell's palsy, lagophthalmos, trichiasis, blepharitis, dacryocystitis, conjunctivitis, corneal dystrophies or degenerations, dry eyes, bullous keratopathy and pre-existing viral keratitis were also noted. Finally the concurrent use of contact lenses and topical steroid was inquired and recorded.
Sample collection: Corneal scrapping samples were taken from 85 patients with clinically suspected keratitis. After a detailed ocular examination using standard techniques, the ophthalmologist took corneal scrapings under aseptic conditions from each ulcer. These samples were labelled with the patient's name and the date of collection. The procedure was performed under high magnification of slit lamp.
After anaes¼thetising the eye with 2-3 drops of sterile proparacaine 0.5% eye drop, cornea was scrapped from the leading edge of the base of the ulcer using fresh sterile Bard-Parker blade no.15. The collected scrapings were then smeared on 2 different slides in a thin, even manner for Gram stain, Giemsa stain and 10% Potassium hydroxide (KOH) wet mount. The use of KOH wet mount lies in dissolving the keratinized epithelial cells that possess a diagnostic difficulty owing to its similarity with fungal strands. The KOH wet mount is very useful for detecting fungi from the ocular specimens.
Inoculation and Incubation: Further scrapings were performed similarly and inoculated directly onto blood agar, chocolate agar, and Sabouraud's dextrose agar in a row of C-shaped streaks. The contact lenses of the patients, if the patient is found to be using, were also cultured. When KOH smears were positive for amoebic cysts further corneal scrapings were performed and the materials were inoculated onto non-nutrient agar. Appropriate steps were taken in consideration with sample collection and transfer to culture media.
All of these culture media inoculated with desired samples was incubated aerobically. The temperature for incubation of inoculated Sabouraud's Dextrose Agar (SDA) was maintained at 27░C for 3 weeks, as well as blood agar and chocolate agar at 37░C for 7 days. These culture media were examined daily for any traces of growth. They were discarded if there were no growths. The growth of fungus in these primary isolated medium was re-cultured in SDA after incubating for 15 days to demonstrate the sporulation. Similarly the temperature for inoculation of non-nutrient agar plates was maintained at 37░C after overlaying with Escherichia coli broth culture. It was examined regularly for the traces of Acanthamoeba species. These culture plates with no signs of growth were also dis-carded after 3 weeks.
Fungal identification: After the subsequent growth and isolation of fungal species, they were identified based on their macroscopic and microscopic features. The microbial cultures were taken as positive when it satisfies at least one of the following crite-ria:
a. Growth and isolation of same organism from more than one solid medium,
b. Confluent growth in one solid medium at inoculated site,
c. Growth and isolation in the medium which is consistent with direct microscopy findings (staining and morphology with Gram-stain),
d. Growth and isolation of the similar organism repeatedly from the same scrapings.
RESULTS- A total of 85 patients with clinically suspected keratitis were examined of which 28 (32.9%) proved to be having fungal infection while the rest of them either had infection with other microorganisms or were culture negative. Out of all the fungal positive cases, 24 (85.7%) had fungal infection alone while 4 (14.2%) had fungus mixed with bacteria. Out of all the fungal positive cases, 26 (92.8%) cases had single species of isolates and remaining 2 (7.1%) cases had two or more species of fungal isolates. Of the total 85 patients included in the study, the ages ranged from 19 to 65 years with 62 (72.9%) being males. Similarly out of the total 28 patients diagnosed with fungal keratitis, the age ranged from 19 to 53 years, with 17 (60.7%) being males.
Out of all the fungus isolated Aspergillus and Fusarium together accounted for 25 (89.28%) cases of which Aspergillus was cultured in 14 (50.0%) while Fusarium was cultured in 11(39.28%) Table 1. Candida was not isolated in any culture.
Table 1: Prevalence of individual fungus isolated from corneal scrapings:
| Type of fungus (species isolated) | No .of cases | N/28 (%) |
|---|---|---|
| Aspergillus spp. | 14 | 50.0% |
| a) Aspergillus fumigatus | 8 | 28.57% |
| b) Aspergillus niger | 6 | 21.43% |
| Fusarium | 11 | 39.28% |
| Penicillium | 2 | 7.14% |
| Sporotrichos | 2 | 7.14% |
| Cephalosporium | 1 | 3.57% |
Trauma was the major cause (n=22, 78.5%) of which vegetative injury was found in 17 (60.7%). Associated history of diabetes mellitus was noted in 5 (17.8%) while of contact lens in 6 (21.4%) patients. Use of corticosteroids predisposing to fungal keratitis was noted in 1 (3.57%) case.
The typical clinical features at presentation in these 28 cases of culture-positive fungal keratitis were as fol-lows: dry, thick and raised corneal surface in 22(78.5%) patients, stromal infiltrates with feathery margins in 19 (67.8%) patients, typical satellite lesions in 3 (10.7%) patients (Fig 1). Hypopyon was present in 15 (53.5%) patients, deep stromal infiltration in 9(32.1%) patients, and corneal abscess in 2 (7.1%) patients at presentation (Fig 2).
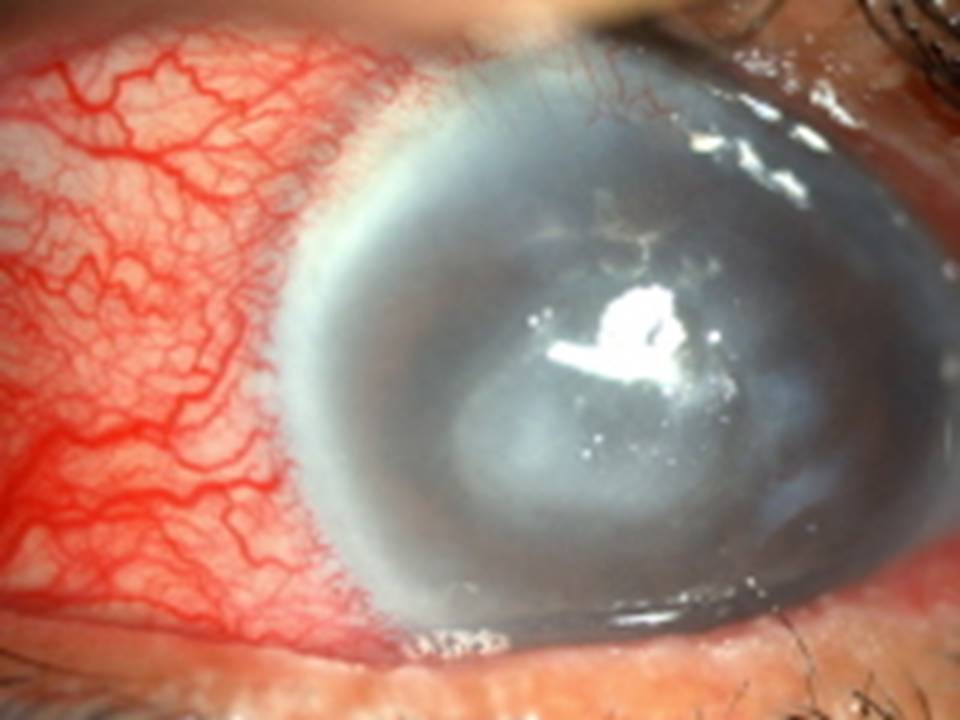
Fig 1: Slit-lamp photo of fungal keratitis with satellite lesions
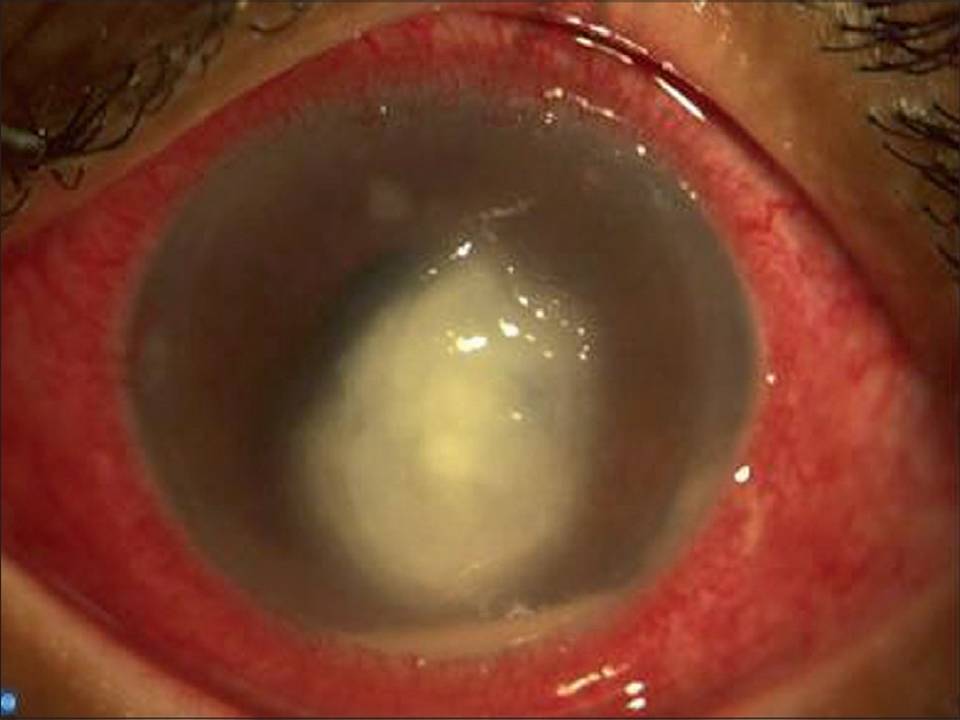
Fig 2: Slit-lamp photo of corneal fungal ulcer with hypopyon
The duration of symptoms of fungal positive cases at presentation ranged from 3 days to 65 days. 13 (46.4%) patients reported cases within 2 weeks, 9 (32.1%) between 3 to 4 weeks, 4(14.2%) between 1 to 2 months and the remaining 2 patients (7.1%) more than 2 months after onset of illness.
DISCUSSION- Contrary to popular perception of low incidence of fungal keratitis in North African region, high incidence (32.9%) of fungal keratitis was found in our study amongst the patient with infectious keratitis. Most of the fungal infections were caused by Aspergillus and Fusarium genera. The higher cases of fungal keratitis in Northern Africa can be attributed to increasing trend towards farming activity in this region which predisposes the people to vegetative injury. As per the prevailing socio-cultural structure, predominantly males go for the outdoor work and the same applies for the farming, hence the male predo-minance in the fungal keratitis.
Although normal cornea is protected against the corneal infections by the constant tear flow maintained by continuous tear secretion and drainage, seasonal variations throughout the year in this region leads to alter tear film. Increasing prevalence of dry eyes is corroborated by the fact that the use of artificial tears has vastly increased in the recent years. Thus the dry eyes hamper the natural defence mechanisms which again could be a major predisposing factor. Due to in-creasing awareness and inclination towards improved cosmetic appearance, the use of contact lenses has increased enormously. Contact lens use is one of the most important factors for infectious keratitis along with ocular and systemic diseases. The overall incidence of contact lens induced keratitis is increasing in the general population.[12] In our study we had patients using contact lenses but none of them were using ReNu mois-ture- loc (Bausch & Lomb) which has been implicated in Fusarium keratitis in different parts of the world [13-14]. Therefore the contact lens related fungal keratitis in our study was independent of the contact lens solution. Hence it could be due to improper handling of contact lenses compiled with lack of awareness of maintaining adequate hygiene.
The use of medications especially steroid eye drops lowers the immune system thereby reducing the host defence mechanism. Steroids are usually prescribed for various ocular diseases like blepharitis, iridocyclitis, choroiditis, and even allergic conjunctivitis. The use of steroids for a spectrum of ocular disorders, if unmonitored, leads to fungal keratitis. Diabetes mellitus was found in some patients as the predisposing factor. Other systemic diseases or autoimmune disorders associated with lowering the immune system or increasing the chances of fungal keratitis were not found in our study.
CONCLUSION- Fungal keratitis is the leading cause of ocular morbidity. Common risk factors include diabetes mellitus, contact lens and corticosteroid use, but traumatic vegetative eye injury being the most common identifiable cause. Due to its varied clinical presentation there is considerable overlap between infectious keratitis and it possesses a clinical challenge for diagnosis. Therefore the knowledge of clinical cases with common causes, associated risk factors and presentations with complications would help in the early diagnosis of fungal keratitis and better clinical outcomes.
REFERENCES
- Collier SA, Gronostaj MP, MacGurn AK, Cope JR, Awsumb KL, Yoder JS, Beach MJ; Centers for Disease Control and Prevention (CDC). Estimated burden of keratitis--United States, 2010. MMWR Morb Mortal Wkly Rep. 2014; 63(45):1027-30.
- Song X, Xie L, Tan X, Wang Z, Yang Y, Yuan Y, Deng Y, Fu S, Xu J, Sun X, Sheng X, Wang Q. A multi-center, cross-sectional study on the burden of infectious keratitis in China. PLoS One. 2014; 9(12):e113843.
- Wang H, Zhang Y, Li Z, Wang T, Liu P. Prevalence and causes of corneal blindness. Clin Experiment Ophthalmol. 2014; 42(3):249-53.
- Wang L, Wang L, Han L, Yin W. Study of Pathogens of Fungal Keratitis and the Sensitivity of Pathogenic Fungi to Therapeutic Agents with the Disk Diffusion Method. J. Curr Eye Res. 2015; 40(11):1095-101.
- Nitulescu C. [Fungal keratitis--diagnostic and therapeutic approach]. [Article in Romanian] Oftalmologia. 2006; 50(4):33-8.
- FlorCruz NV, Evans JR. Medical interventions for fungal keratitis. Cochrane Database Syst Rev. 2015; (4):CD004241.
- Garg P, Roy A, Roy S. Update on fungal keratitis. Curr Opin Ophthalmol. 2016; 27(4):333-9.
- Barut Selver O, Egrilmez S, Palamar M, Arici M, Hilmioglu Polat S, Yagci A. Therapeutic Corneal Transplant for Fungal Keratitis Refractory to Medical Therapy. Exp Clin Transplant. 2015;13(4):355-9.
- Huang JF, Zhong J, Chen GP, Lin ZT, Deng Y, Liu YL, Cao PY, Wang B, Wei Y, Wu T, Yuan J, Jiang GB. A Hydrogel-Based Hybrid Theranostic Contact Lens for Fungal Keratitis. ACS Nano, 2016, 10 (7), pp 6464¢6473.
- Reddy JC, Murthy SI, Reddy AK, Garg P. Risk factors and clinical outcomes of bacterial and fungal scleritis at a tertiary eye care hospital. Middle East Afr J Ophthalmol. 2015; 22(2):203-11.
- Gupta MK, Chandra A, Prakash P, Banerjee T, Maurya OP, Tilak R. Fungal keratitis in north India; Spectrum and diagnosis by Calcofluor white stain. Indian J Med Microbiol. 2015; 33(3):462-3.
- Lin TY, Yeh LK, Ma DH, Chen PY, Lin HC, Sun CC, Tan HY, Chen HC, Chen SY, Hsiao CH. Risk Factors and Microbiological Features of Patients Hospitalized for Microbial Keratitis: A 10-Year Study in a Referral Center in Taiwan. Medicine (Baltimore). 2015; 94(43):e1905.
- Centers for Disease Control and Prevention (CDC). Fusarium keratitis--multiple states, 2006. MMWR Morb Mortal Wkly Rep. 2006; 14;55(14):400-1.
- Epstein AB. In the aftermath of the Fusarium keratitis outbreak: What have we learned? Clinical ophthalmology (Auckland, NZ). 2007; 1(4):355-366.
| Source of Financial Support: Nil Conflict of interest: Nil |
Ā
|---|
| International Journal of Life-Sciences Scientific Research (IJLSSR) Open Access Policy Authors/Contributors are responsible for originality, contents, correct references, and ethical issues. IJLSSR publishes all articles under Creative Commons Attribution- Non-Commercial 4.0 International License (CC BY-NC). https://creativecommons.org/licenses/by-nc/4.0/ |
Ā
|---|