Int. J. Life. Sci. Scienti. Res., 3(6):
1495-1499,
November 2017
Chemical Characterization
and Larvicidal Activity of Essential Oil from Aniba duckei Kostermans against Aedes
aegypti
Rogerio De Mesquita Teles1*, Victor Elias Mouchrek Filho2, Antonio
Gouveia De Souza3
1 Federal
Institute of Education Science and Technology of Maranhao,
Chemistry Academic Department Campus Sao
Luis- Monte Castelo, Sao Luis- MA, Brazil
2Federal
University of Maranhao, Chemical Technology
Department, Sao Luis- MA, Brazil
3Federal
University of Paraiba, Chemistry Department, Joao Pessoa– PB, Brazil
*Address for
Correspondence: Dr. Rogerio De Mesquita Teles, Teacher, Department of Chemistry Academic, Federal
Institute of Education, Science and Technology of Maranhao,
Sao Luis Campus- Monte Castelo, Getulio
Vargas Avenue 04, CEP 65030-005, Sao Luis- MA, Brazil
Received: 13 July 2017/Revised: 23
August 2017/Accepted: 19 October 2017
ABSTRACT- Aedes aegypti mosquito is the major
vector of zika, chikungunya,
and dengue fever. These diseases incidence has been growing rapidly in many
points of the globe in the past few years. And because there’s no vaccine for
them yet, the best way to fight those diseases is to attack their vector, specially by eliminating potential sites for its oviposition and larvae growth. Nowadays, organophosphorus insecticides are used in increasing doses,
which targets Aedes aegypti
resistant populations. Aniba duckei Kostermans, which is
known as rosewood and belongs to the Lauraceae family,
is a species with trees up to 30 meters tall and 1 meter in trunk diameter. It
is essential oil used in perfumery due to its high content of linalool. This
research identified the components of essential oil from Aniba duckei Kostermans
thin branches and leaves and then applied it as larvicide
against Aedes aegypti,
and its effects were measured by calculation of concentration at which half
larvae die (LC50). Average yield found for oil by plant was 1.93% by
mass. The major component in rosewood essential oil is linalool, whose
concentration was found 89.34% by mass. LC50 for the essential oil
was 250.61 (±2.20) µg mL-1, for l-linalool, 279.89
(±2.12) µg mL-1, and for dl-linalool was 346.73 (±2.14) µg mL-1.
Key-words-
Essential oil, Aniba duckei
Kostermans, Linalool, Aedes
aegypti, Larvicide
INTRODUCTION- The world has experienced a dengue incidence increase in the last
50 years. Recent studies estimate about 395 million cases of dengue hemorrhagic fever in 100 countries, of which 500
thousand are classified as dengue hemorrhagic fever/ dengue
shock syndrome (DHF / DSS) [1]. Disease is caused by four
serotypes of dengue virus, DENV-1, DENV-2, DENV-3 and DENV- 4 [2].
This is the most important arbovirosis worldwide with about 50 million infections per
year [3], and it can be asymptomatic or
manifest many symptoms, from self-limited febrile illness to severe forms that
may lead to death [4].
In terms of morbidity and
mortality, dengue is nowadays considered the most important viral disease
transmitted by mosquitoes, constituting a serious public health problem of
urban centers from South and Central America, Southeast Asia and West Pacific
tropical areas [5].
Chikungunya disease, which shown
symptoms similar to dengue’s is caused by Chikungunya virus (CHIKV), a RNA virus member of the Alphavirus genus in the family Togaviridae,
first described in Africa, but which migrated later to Asia and Europe, after
small mutations [6-8]. These disease symptoms,
which may persist for months or even years, are debilitating, causing fever, arthralgia or severe arthritis and itchy skin [9].
Zika virus is a
flavivirus (Flaviviridae
family) originally isolated in Uganda, in 1947 [10]. From 1951
to 2013, serological evidence in humans were notified
in African countries (Uganda, Tanzania, Egypt, Central African Republic, Sierra
Leone and Gabon), Asian countries (India, Malaysia, Philippines, Thailand,
Vietnam and Indonesia) and Oceanian countries
(Micronesia and French Polynesia). In the Americas, zika
virus was identified in Easter Island, Chile’s territory in the Pacific Ocean
which is 3.500 km from the mainland, only in the beginning of 2014 [11].
Since May
2015, Brazil’s Ministry of Health has been registering cases of zika virus in the country [12]. Usually,
infection is characterized by fever, skin rash, joint pain or conjunctivitis,
that may last for days or weeks, and its symptoms are many times confused with
dengue’s or chikungunya’s, which may result in
diagnostic errors [13].
Dengue, chikungunya and zika are all
transmitted by the same vector, Aedes aegypti mosquito [8,10,14-15].
Because there are still no validated vaccines against dengue or a
specific antiviral for treatment of those diseases [16-18], the best
control method is prevention, by attacking its vector [19]. Vector
control is done by eliminating propitious
locations for oviposition or by fighting these
mosquito larvae. In recent times, this combat has been carried out by
applications of organophosphorus insecticides in
increasing doses, which has caused mosquitoes to become resistant to pesticides
[20-21].
Plants that are source of
molecules with phage inhibitory, repellent and insecticidal actions, in
addition to substances that are able to change growth regulation, are a good
alternative to the use of insecticides. Essential oils, produced in the
secondary metabolism of plants, have also been shown to be a good source of
materials with insecticidal, larvicidal and repellent
action [15,22-25].
Botanical species Aniba duckei Kostermans, of Lauraceae family [26-27],
synonym of Aniba rosaeodora Ducke [28-30], has many common names, like: pau-rosa, pau-rosa-do-amazonas and umbaúba (Brazil),
rosewood (English speaking countries), bois de rose femelle
(French Guyana), enclit rosenhout
(Suriname), cara-cara (Guyana) [30] and palo de rosa, in Castilian speaking Amazonian countries [31].
Linalool (3,7-Dimethyl-1,6-octadien-3-ol), shown in Fig. 1 is the major
component of Aniba duckei Kostermans essential oil [32]. Other minor
components are also present in this essential oil’s composition.
Linalool,
which is an alcoholic monoterpene
and one of the most important substances for fragrance industry [33],
occurs naturally as two stereoisomers,
3R-(-)-linalool and 3S-(+)-linalool [34]. Fig. 1 (A and B) below has
shown the structures for linalool.
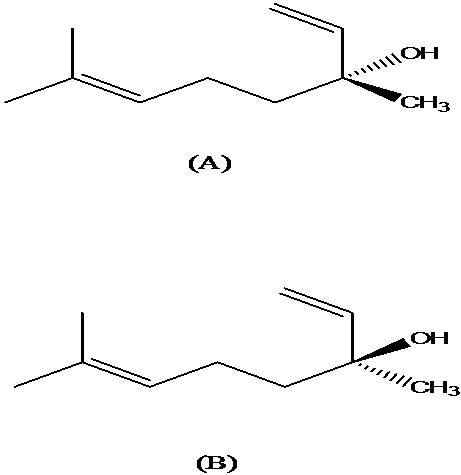
Fig. 1: Enantiomeric structures for linalool: (A) 3R-(-)-linalool
or lincareol; (B) 3S-(+)-linalool or coryandrol
To contribute in the fight
against Aedes aegypti larvae, the
essential oil from Aniba duckei
Kostermans was extracted, and then its physical-chemical properties were
evaluated, as well as its larvicidal activity against larvae of the Aedes
aegypti mosquito in third or fourth stages.
MATERIALS AND METHODS-This research
was developed in the Laboratory of Fuels and Materials (LACOM) located at
Paraiba Federal University (UFPB) in partnership with Laboratory of Analytical
Chemistry (LPQA), Analytical Center and Laboratory of Physical Chemistry,
Microbiology of the Technological Pavilion of the Federal University of
Maranhao (UFMA), Laboratory of Researches and Tests of Fuels (LAPEC) of the
Federal University of Amazonas (UFAM) and São Carlos Institute of Chemistry
from University of Sao Paulo (USP).
Samples, leaves and thin
branches, collected from three Aniba duckei Kostermans trees cultivated
in the Ducke Forest Reserve, highway AM–010, 26 km, Manaus, Amazon, Brazil
(03º00''02'' and 03º08''00'' south latitude and 59º58'00'' west longitude),
were dried for seven days under natural ventilation and then crushed. Essential
oil was extracted from 30 grams of thin branches with 300 mL of distilled
water, by hydro distillation using Clevenger system, under the temperature of
100°C. After that, the oil was dried by percolation in anhydrous Na2SO4
and then stored in glass ampoules under refrigeration.
Yield, density, extraction time,
ethanol solubility, refractive index, oil extraction yield, color and
appearance were determined. As standards were used racemic linalool from
Aldrich (Aldrich Chemical Co) and R-(-)-linalool from Fluka (Fluka Chemie
GmbH). Standard solutions of monoterpenes in ethanol and in hexane were
prepared by dilution at different concentrations.
GC-MS
essential oil analysis was performed on a Varian chromatograph, model 3900,
using helium as carrier gas with flow in the column of 1 mL min-1;
Injector temperature: 270°C, split 1:50; capillary column (30 mx 25 mm) by
stationary phase VF-1ms (100% methylsiloxane 0.25 μm) and oven temperature
programmed to 60°C and then increased to 220°C at a rate of 4°C min-1 and then
increased again to 260°C, this time at a rate of 1°C min-1, with total running
time of 100 minutes. For mass spectrometer, the manifold, ion trap and transfer
line temperatures were set to 50, 190 and 200°C, respectively. 1.0 μL
(automatic injector CP-8410) aliquots of the samples diluted were injected in
proportion of 20 μL for 1.5 mL of hexane. Linalool was quantified by the
external standard method, considering its high concentration in the samples.
In order to collect Aedes
aegypti eggs, a simple trap was prepared using 500 ml plastic jars
half-filled with water and a piece of wood of approximately 20 cm x 5 cm with one
part submerged. For hatching, the eggs were immersed in a plastic container
with 3 liters of mineral water and 500 milligrams of rat feed. After immersion
of the eggs, 0.5 g more of rat feed was added, to aid in larvae growth. All
material was kept inside a wooden cage and was covered with a fabric screen,
suitable for insects, in order to avoid contamination by eggs of other
mosquitoes’ species. After hatching, the larvae were monitored until they
reached the 3rd or 4th stage of development, from 4 to 5
days, when they were then used in the larvicidal activity tests.
For toxicity test, 10 larvae were transferred to a beaker
containing 20 mL of mineral water (26-28°C). Each test was carried out five
times for each concentration tested. Positive controls were performed with the
organophosphate temephos in Aedes aegypti larvae at the concentration
used by the sanitary surveillance which is 100 ppm. Negative controls were
performed with 20 mL mineral water (26 - 28 ° C) containing 0.04% Tween. Larvae
were exposed to the solutions for 24 hours and at the end of this period
mortality was recorded.
STATISTICAL ANALYSIS - Statistical analysis of data was performed according to the
Reed-Muench method by plotting the mortality data for each concentration
tested, where one curve is observed for accumulation of dead animals at each
concentration and another one for accumulation of survivors. The point of
intersection between the curves is the median lethal concentration (LC50),
because at this point the number of surviving animals is equal to the number of
dead animals [35]. Confidence interval was calculated according to
the PIZZI method [36]
RESULTS AND DISCUSSION- The extraction
time with the best yield was obtained after four hours of extraction, yielding 1.87%
(m/m). Density, 70% (v/v) ethanol solubility, and refractive index for this
essential oil were respectively 0.86 g / mL, 1:2 and 1.46. These data, together
with the yellow color and clear appearance observed, are in agreement with
literature data [37].
The substances identified
from the chromatogram are listed in Table 1. For identification of the
compounds were used the spectral databases of the spectral libraries NIST105,
NIST21 and WILEY139, and AMSDIS (Automated Mass Spectral Deconvolution Mass &
Identification System) software, as well as references [38]. For
linalool, confirmation was also by addition of standard.
Table
1: Identified compounds in a sample of essential oil from Aniba duckei Kostermans’ branches
|
Pico |
tRETa |
Compound Name |
%Ab |
|
1 |
15.61 |
Limonene |
0.52 |
|
2 |
15.71 |
1,8-Cineole |
1.07 |
|
3 |
17.43 |
Cis-linalool oxide |
1.94 |
|
4 |
18.06 |
Trans-linalool oxide |
1.86 |
|
5 |
18.60 |
Linalool |
89.34 |
|
6 |
21.88 |
α-Terpineol |
3.06 |
|
7 |
28.26 |
α-Copaene |
0.89 |
|
8 |
31.74 |
α-Patchoulene |
0.77 |
|
9 |
32.02 |
Caryophyllene |
0.55 |
a = Peak
retention time by column elution order.
%Ab = normalized area percentage.
From the graph
it’s possible to see linalool, C10H18O, as the major component,
with 89.34%, followed by α-terpineol, C10H18O, whose
area percentage was 3.06%.
Larvicidal activity of essential oil from Aniba
duckei Kostermans was tested in seven concentrations: 100, 150, 200, 250,
300, 350 and 400 μg mL-1, with 10 larvae used for
each concentration. The tests were performed five times for each concentration
and data on the number of live and dead larvae were obtained by an average of
the five replicates.
For linalool (dl-linalool and l-linalool)
standards, major component of the essential oil from Aniba duckei
Kostermans, larvicidal activity was tested at the same seven concentrations at
which the essential oil was tested, also five times for each concentration. The
results are summarized in Table 2.
Table
2: Estimation of LC50 of essential oil and linalool (dl-linalool and
l-linalool) by Reed-Muench method based on accumulation of dead and live larvae
|
Doses (µg mL-1) |
Log dose |
Mortality (%) |
||
|
Oil |
dl-linalool |
l-linalool |
||
|
400 |
2.60 |
100 |
66.0 |
100 |
|
350 |
2.54 |
76 |
38.7 |
100 |
|
300 |
2.48 |
56 |
28.0 |
44 |
|
250 |
2.40 |
40 |
16.0 |
34 |
|
200 |
2.30 |
34 |
6.0 |
18 |
|
150 |
2.18 |
30 |
0.0 |
4 |
|
100 |
2.0 |
18 |
0.0 |
0 |
L-linalool killed 100% of the larvae at lower
concentrations, from 350 µg mL-1, where the oil alone has only
reached 100% at 400 µg mL-1 and dl-linalool has not reached this
level at the analyzed concentration range. When investigating median lethal
concentration (LC50), the best larvicidal activity was detected for
the essential oil from Aniba duckei Kostermans, LC50 = 250.61
(± 2.20) µg mL-1, against LC50 of 279.89 (± 2.12) µg mL-1
of l-linalool and LC50 = 346.73 (± 2.14) µg mL-1 for
dl-linalool. Thus, it is concluded that the linalool responsible for larvicidal
activity should be the levorotatory isomer (l-linalool). No information was
found though, in the literature data, on larvicidal activity against Aedes
aegypti for l-linalool, whereas for dl-linalool, the results obtained are
in accordance with the literature data, which does not attribute to linalool a
value of larvicidal activity, but to the interval greater than 100 µg L‑1
(> 100 µg L‑1) [39].
CONCLUSIONS- In this
research, essential oil from Aniba duckei
Kostermans presented 1.87% (m/m) extraction yield, with linalool being its
major component (89.34%), followed by
α-terpineol (3.06%). The best result of median lethal concentration (LC50)
against Aedes aegypti was
the one for the essential oil, followed by the results for l-linalool, which is
responsible for linalool’s larvicidal. Once essential oil is a natural product
and, therefore, less harmful to humans’ and domestic animals’ health, it can be
used as a larvicide in at larval growth sites of Aedes aegypti in order
to reduce the impact of synthetic insecticides on health of people and the
environment. Besides, the complex composition of the essential oil makes it
harder for mosquitoes to develop resistance. Other advantages of essential oil
from Aniba duckei Kostermans discovered during this research includes
environmental, economic and social aspects, since the oil is prepared using
just leaves and thin branches from reforested plants, its final cost is low
compared to synthetic insecticides’ and it also can generate jobs and income to
local residents, from production to commerce.
REFERENCES
1. Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL,
Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF, George DB, Jaenisch T, Wint
GR, Simmons CP, Scott TW, Farrar JJ, Hay SI. The global distribution and burden of
dengue. Nature, 2013; 496:504–507.
2. Dos Santos TP, Cruz OG, da Silva KAB, de
Castro MG, de Brito AF, Maspero RC, de Alcântra R, Dos Santos FB, Honorio NA,
Lourenço-de-Oliveira R. Dengue serotype circulation in natural populations of Aedes
aegypti. Acta
Tropica, 2017; 176:140–143.
3. Viana DV, Ignotti E. The occurrence of dengue and weather changes
in Brazil: a systematic review. Revsta Brasileira de Epidemiologia, 2013; 16(2):240-256.
4.
Dias LBA, Almeida SCL, de Haes TM, de Mota LM, Roriz-Filho JS. Dengue: transmission, clinical aspects, diagnosis and treatment.
MEDICINA-Ribeirão Preto, 2010; 43(2):143-52.
5. Brazil Ministry of Health –
National Health Foundation (FUNASA). (2002). Programa Nacional de Controle da Dengue. Retrieved from the Virtual
Health Library from Ministry of Health website:
http://bvsms.saude.gov.br/bvs/publicacoes/pncd_2002.pdf.
6.
Weaver SC. Arrival of chikungunya virus in the new
world: prospects for spread and impact on public health. PLOS Neglected
Tropical Diseases, 2014; 8(6):
2921.
7.
Powers AM, Logue CH. Changing patterns of chikungunya virus:
re-emergence of a zoonotic arbovirus. Journal of General Virology, 2007; 88(9):2363-2377.
8.
Madariaga M, Ticona E, Resurrecion C. Chikungunya: bending over the Americas and
the rest of the world. The Brazilian
Journal of Infectious Diseases, 2015; 20(1):91-98.
9. Yang S, Fink D, Hulse AR, Pratt D. Regulatory considerations in development
of vaccines to prevent disease caused by Chikungunya virus. Vaccine, 2017;
35(37):4851-4858.
10.
Wikan N. Suputtamongkol Y,
Yoksan S. Smith, D. R. Immunological
evidence of Zika virus transmission in Thailand. Asian Pacific Journal of Tropical Medicine, 2016; 9(2):141-144.
11.
Oswaldo Cruz Foundation
(Fiocruz). Zika.
Retrieved from the Oswaldo Cruz Foundation website: https://agencia.fiocruz.br/zika.
12.
Calvet GA, Filippis AMB,
Mendonça MC, Sequeira PC, Siqueira AM, Veloso VG, Nogueira RM, Brasil P. First detection of autochthonous Zika virus
transmission in a HIV-infected patient in Rio de Janeiro, Brazil. Journal of Clinical
Virology,
2016; 74:1-3.
13. Frankel MB, Pandya K, Gersch J, Siddiqui
S, Schneider GJ. Development of the Abbott Real Time ZIKA assay for the
qualitative detection of Zika virus RNA from serum, plasma, urine, and whole
blood specimens using the m2000 system. Journal of Virological Methods, 2017;
246:117–124.
14. Riou J, Poletto C, Boëlle P-Y. A
comparative analysis of Chikungunya and Zika transmission. Epidemics, 2017;
19:43–52.
15.
Costa JGM, Rodrigues FFG, Angelico EC, Silva MR, Mota ML, Santos NKA,
Cardoso ALH, Lemos TLG. Chemical-biological study of the essential oils of Hyptis
martiusii, Lippia sidoides and Syzigium aromaticum against larvae
of Aedes aegypti and Culex quinquefasciatus. Brazilian Journal of Pharmacognosy, 2005; 15(4):304-309.
16. Kang DS, Alcalay Y, Lovin DD, Cunningham JM, Eng MW, Chadee DD, Severson DW. Larval stress
alters dengue virus susceptibility in Aedes aegypti (L.) adult females. Acta Tropica, 2017; 174:97–101.
17.
Tabanca N, Demirci B, Ali A, Ali Z, Blythe EK, Khan IA. Essential
oils of green and red Perilla frutescens as potential sources of
compounds for mosquito management. Industrial
Crops and Products, 2015; 65:36–44.
18.
Paiva MHS, Lovin DD, Mori A, Maria AV, Santos MAM, Severson, DW, Ayres
CFJ. Identification of a major Quantitative Trait Locus
determining resistance to the organophosphate temephos in the dengue vector
mosquito Aedes aegypti. Genomics,
2016; 107(1):40–48.
19. Govindaranja M, Sivakumar R, Rajeswary M, Yogalakshmi K. Chemical,
composition and larvicidal activity of essential oil from Ocimum basilicum
(L.) against Culex tritaeniorhynchus, Aedes albopictus and Anopheles
subpictus (Diptera:Culicidae). Experimental Parasitilogy,
2013; 134(1):7-11.
20. Lima JB, Da Cunha MP, Da Silva RC, Galardo
AK, Soares S da S, Braga IA, Ramos RP, Valle D. Resistance of Aedes aegypti to organophosphates in
several municipalites in states of Rio de Janeiro and Espirito Santo, Brazil. The American Journal of Tropical Medicine
and Hygiene, 2003; 68(3):329-333.
21. Braga IA, Pereira JBL, Soares S da S,
Valle D. Aedes aegypti resistance to
temephos during 2001 in several municipalities in the states of Rio de Janeiro,
Sergipe and Alagoas, Brazil. Memórias
do Instituto Oswaldo Cruz, 2004; 99(2):
199-203.
22.
Fujiwara GM, Annies V, de Oliveira CF, Lara RA, Gabriel MM, Betim FC,
Nadal JM, Farago PV, Dias JF, Miguel OG, Miguel MD, Marques FA, Zanin SM. Evaluation of larvicidal activity and ecotoxicity of linalool,
methyl cinnamate and methyl cinnamate/linalool in combination against Aedes
aegypti. Ecotoxicology and Environmental Safety, 2017; 139:238-244.
23.
Saavedra L, Romanelli MGP, Rozo CE, Duchowicz PR. The quantitative structure–insecticidal activity relationships
from plant derived compounds against chikungunya and zika Aedes aegypti
(Diptera:Culicidae) vector. Science of the Total
Environment, 2017; 610-611:937-943.
24.
Carrasco H, Raimondi M, Svetaz L, Di Liberto M, Rodriguez MV, Espinoza
L, Madrid A, Zacchino S. Antifungal activity of eugenol analogues. Influence of different substituents and studies on mechanism of
action. Molecules, 2017; 17(1):1002–1024.
25.
Murugan K, Murugan P,
Noortheen A. Larvicidal and repellent potential of Albizzia amara Boivin and Ocimum
basilicum Linn against dengue vector, Aedes
aegypti (Insecta: Diptera:Culicidae). Bioresource Technology, 2007; 98(1):198–201.
26. Correa DB, Gottilieb OR. Duckein, an alkaloid from Aniba duckei.
Phytochemistry, 1975;
14(1): 271-272.
27. Siani AC, Sampaio ALF, de Sousa MC,
Henriques MGMO, Ramos MFS. Essential oils–
anti-inflammatory potential. Revista Biotecnologia Ciência & Desenvolvimento, 2000; 16:38-43.
28. Ducke A. Aromatic Lauraceae of Amazon. South American Botany Meeting, 1938;
3:55-74.
29.
Sampaio PTB, Barbosa AP, Vieira G, Spironello WR, Bruno FMS. Canopy sprouting biomass of rosewood (Aniba rosaeodora
Ducke) in an Amazonian terra firme forest. Acta Amazonica, 2005; 35(4):491-494.
30.
Maia JGS, Zoghbi MGB, Andrade EHA. Aromatic
plants in Amazon and their essential oils. 1st ed., Brazil; Emilio
Goedi Paraense Museum: 2002; pp:173.
31.
Clay JW, Clement CR.
Selected species and strategies to enhance income generation from Amazonian
forests. Retrieved from the Food and
Agriculture Organization of the United Nation website: http://www.fao.org/docrep/v0784e/v0784e00.htm.
32.
Maia JGS, Maia, Mourão, R H.
V. (2015) Amazon Rosewood (Aniba
rosaeodora Ducke) Oils. In. V. Preedy (Ed.), Essential Oils in Food Preservation, Flavor and Safety (pp.
193–201). San Diego, CA: Academic Press.
33.
Vatanparast J, Bazleh S, and
Janahmadi M. The effects of linalool on the excitability of central neurons of
snail Caucasotachea atrolabiata. Comparative Biochemistry and
Physiology, 2017; 192:33-39.
34.
Siani AC, Monteiro SS, Garrido IS, Ramos MCKV, Aquino-Neto FR. Chemical variability of linalool in the essential oil of Aeollantus
suaveolens (Lamiaceae). Fitos, 2005; 1(2):59-63.
35.
Colegate SM, Molyneux RJ.
Bioactive Natural Products: Detection, Isolation, and Structural Determination.
1st ed., England, CRC Press:
1993; pp:528.
36. Pizzi M. Sampling variation of the fifty percent end-point,
determined by the Reed-Muench (Behrens) method. Human Biology, 1950; 22(3):151-190.
37. Raoul D. Etude biographique et critique. Genebra;
Skira: 1953; pp:120.
38.
Adams RP. Identification of
essential oil components by gas chromatography/mass spectroscopy. 3rd
ed., USA; Allured Publishing Corporation: 2001; pp:800.
39.
Simas NK, Lima EC, Conceição SR.. Natural
products for dengue transmission control- larvicidal activity of Myroxylon
balsamum (red oil) and of terpenoids and phenylpropanoids. Quimica Nova, 2004; 27(1):46-49.
40. Augusto LGS, Freitas CM. The Principle of Precaution in the use indicators of environmental
chemical risks to occupational health. Journal of Science & Collective
Health,
1998; 3(2):85-95.
41.
Augusto LGS, Câmara-Neto HF. (2007)
Dengue: unsustainability of PEAa. In XXVII Inter- American Congress of Sanitary
and Environmental Engineering (pp. 1-6). Porto Alegre.